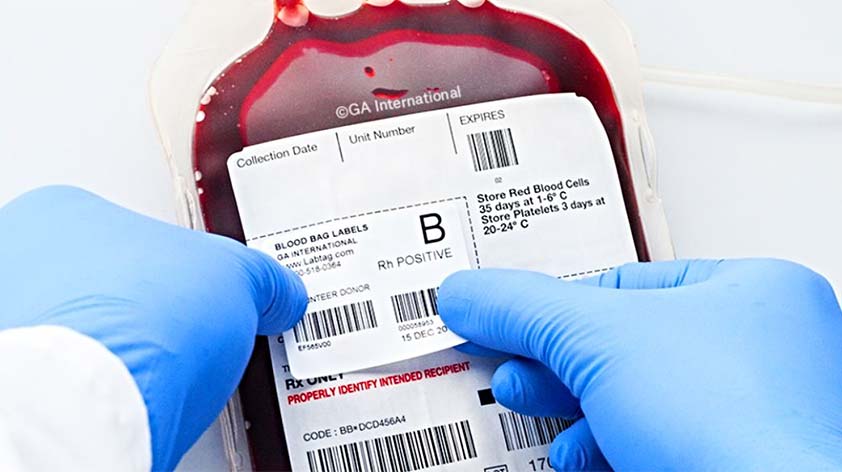
Blood bag identification is a crucial aspect of blood transfusion processes, ensuring the safe and accurate delivery of blood products to patients in need. However, with multiple blood bag manufacturers and differing naming conventions, navigating the identification rules can be a complex and daunting task.
In order to ensure consistency and clarity in blood bag identification, several organizations, including the International Society of Blood Transfusion (ISBT) and the Food and Drug Administration (FDA), have established guidelines and regulations. Despite these efforts, discrepancies and confusion still arise, leading to potential errors and delays in patient care. Follow on for Blood Bag Identification: Navigating the Naming Rules.
Understanding the importance of accurate blood pouch labelling
In discussing the importance of standardization and the potential solutions for improving blood bag identification processes – the ultimate goal is of promoting patient safety and efficient transfusion practices. It is imperative for healthcare professionals to have a thorough understanding of blood bag naming rules and their implications.
In the medical field, accurate labeling and identification of blood bags is crucial for patient safety. A single mistake in identifying a blood bag could result in a life-threatening transfusion error. This is why organizations like ISBT and FDA have established guidelines for blood bag labeling, to ensure consistency and minimize the risk of errors.
However, despite these efforts, discrepancies and confusion still occur due to the multiple manufacturers and naming conventions present in the industry. It is important for healthcare professionals to understand the importance of accurate blood bag labeling and the potential consequences of not following standardized practices.
By improving communication and implementing standardized labeling practices, we can work towards minimizing errors and improving patient care.
Compliance with naming conventions: essential guidelines
In order to ensure proper labeling and identification of blood bags, healthcare professionals must comply with naming conventions that have been established by regulatory bodies. These guidelines outline specific requirements for labeling elements such as product name, lot number, and expiration date.
It is essential that these guidelines are followed in order to avoid confusion and potential errors in patient care. By adhering to these naming conventions, healthcare professionals can ensure the safe and accurate use of blood bags in medical settings.
Key components of blood bag identification
One of the key components of blood bag identification is the use of unique identification numbers. Each blood pouch must have a unique number assigned to it, which is used to track its journey from the donor to the recipient. This number is typically a combination of letters and numbers, and it is essential that it is legible and accurate to prevent any mix-ups. Another crucial component is the inclusion of important information, such as the blood type and expiration date, on the blood bag label.
This information allows healthcare professionals to quickly and easily identify the contents of the bag and ensure that it is suitable for the intended recipient. Additionally, proper labeling with the donor’s name and identification number is necessary for traceability and to prevent any potential allergic reactions. Overall, understanding and correctly implementing the key components of blood bag identification is essential for safe and effective use of blood products in healthcare settings.
Navigating naming rules: common challenges
However, navigating the naming rules for blood bag identification can present some common challenges. One challenge is ensuring consistency and accuracy in labeling, especially when multiple blood products are being used at the same time. Another challenge is keeping up with changing regulations and guidelines, as naming rules for blood pouch identification can vary between different countries and regions.
Additionally, human error can also play a role in incorrect labeling, emphasizing the need for thorough training and quality control measures. Overcoming these challenges is essential for maintaining safe and efficient blood bag identification practices in healthcare facilities.
Best practices for proper blood bag identification
It is imperative for healthcare facilities to establish and follow best practices for proper blood pouch identification. This includes implementing consistent and accurate labeling techniques, regularly reviewing and updating naming protocols according to changing regulations, and adopting measures to prevent human error.
By adhering to these practices, healthcare professionals can ensure the safe and efficient identification of blood pouches, reducing the risk of potential errors and ensuring patient safety. In addition, maintaining proper blood bag identification practices can also improve workflow and efficiency in healthcare facilities, ultimately leading to better patient outcomes.
In conclusion, understanding the complex naming rules for blood bags is crucial in maintaining safety and efficiency in healthcare facilities. By adhering to the guidelines set by governing bodies such as the FDA and AABB, medical professionals can ensure that blood products are accurately identified and used for their intended purposes.
It is also important for healthcare organizations to stay updated on any changes or updates to these rules in order to provide the best possible care for their patients. With proper knowledge and adherence, we can continue to navigate the world of blood bag identification with confidence and accuracy.